AGuIX® at a glance
The goal of AGuIX® is to improve the efficacy of radiotherapy of cancer
leading to a better survival and quality of life
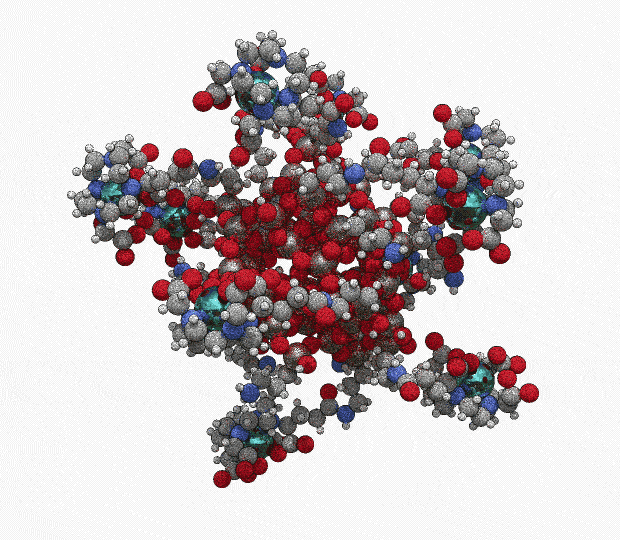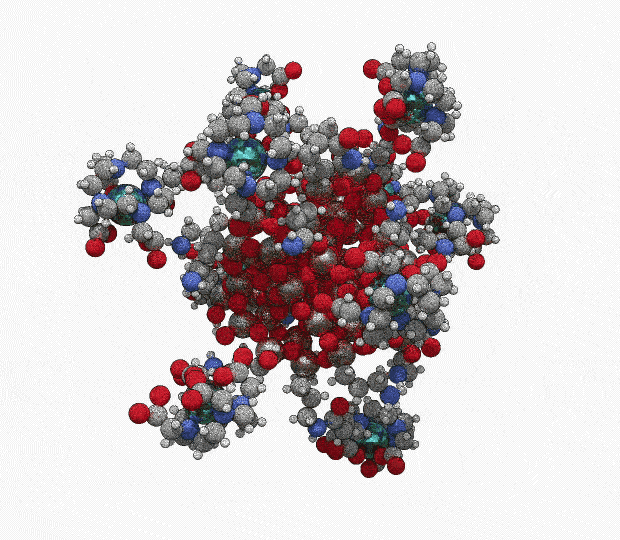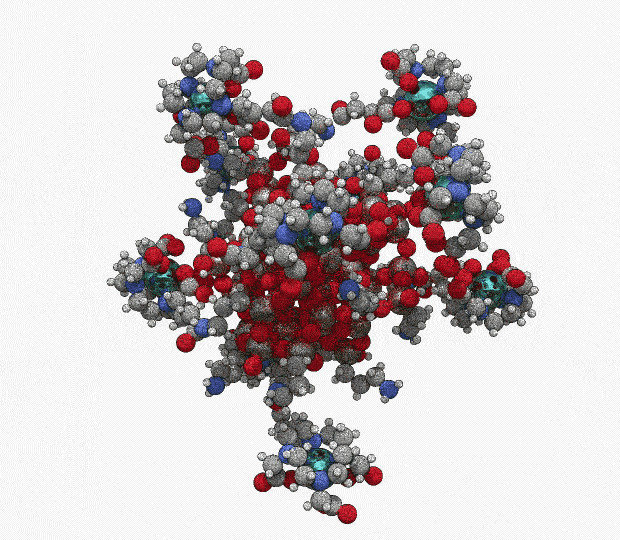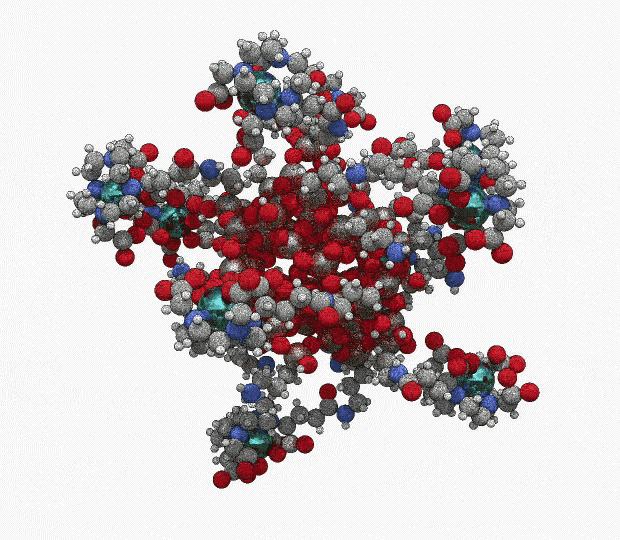
AGuIX® concentrates a high number of gadolinium atoms (∼15) in an ultrasmall size object (∼5 nanometers)
These key structural attributes lead to a unique combination of three pharmacological properties that can be translated into clinical benefits
The ultrasmall size of the structure allows the selective accumulation of AGuIX® in tumors after intravenous administration
The presence of gadolinium in the structure provides contrast imaging property to AGuIX® (positive contrast agent for MRI – Magnetic Resonance Imaging)
The high concentration of gadolinium within the structure brings the ability of AGuIX® to enhance the radio-induced damages in tumors: radiosensitizing effect




